|
The question says: What is the molar mass of calcium phosphate? Agree?īut, what if you have to deduce the formula first before calculating the molar mass? Here is what I mean. When you are given the formula, things become much easier. So this was the simple concept about these conversions. The formula is: Molar mass = mass / moles Q: What is the molar mass of CH4 (methane) if there are 0.623 moles in a 10 g sample? To do so, we are simply going to rearrange the above formula: But, how can you calculate the molar mass using moles? So this is how you can use mass and molar mass to calculate moles. How many moles are present in 90 g of NaOH? Q: The molar mass of sodium hydroxide (NaOH) is 40 g/mol. Moles = (Mass of the substance) / (Molar mass)Ĭovalent Bonding of water (H2O) | Must Read To convert molar mass to moles for a substance, we divide the mass of the substance by its molar mass. In simple words, it is the SI unit (International System of Units) for the amount of substance. Mole: A mole is a mass of a substance that consists of 6.023 x 10 23 particles of the substance. But first of all, let me introduce moles to you. When studying this topic, it is important to learn this calculation. Finally, we add all the values to get the molar mass in g/mol. Then, we apply the formula: (A r of the element x number of atoms of the element). Then, we find the Ar values of the elements. For example, the elements in AgCl are silver and chlorine. First of all, we identify the elements in the compound. Summary: To find the molar mass in periodic table, we do four steps. : Here are the A r values of these two elements: When we know this, it is time to move on to the second step. : What elements are present in aluminium oxide? After looking at the formula, you will say:Īluminium and oxygen. The molar mass of Aluminium oxide ( Al 2O 3).: Now, we are going to add these values to get the molar mass of silver nitrate (AgNO3) : Now, we are going to use this formula: (A r of the element x number of atoms of the element). : Here are the Ar values of these elements: With this, it is time to move on to the second step. We have silver (Ag), nitrogen (N) and oxygen (O). : From the formula, can you find out what elements are present in silver nitrate? The molar mass of silver nitrate (AgNO 3).Now, let’s take a look at some other examples as well. : Finally, you are going to add all the values to get your molar mass. In simple words, we are going to do this: (A r of the element x number of atoms of the element). :max_bytes(150000):strip_icc()/GettyImages-175532236-c614b233b7e84d5487cad8b280f365a4.jpg)
So now, we are going to multiply the number of atoms with their Ar values that we found out in step 2. Moreover, sulfur has only one atom (shown by S), and oxygen has four atoms (shown by O4). Hydrogen has two atoms (suggested by H2). : From the formula (H2SO4), we can say the following: When you have used the periodic table to calculate the Ar values, move on to the third step. : Now, let’s use the periodic table to find the Ar values of these elements. From the formula H2SO4, we can say that this compound has three elements: : First of all, we will identify all the elements in this compound. 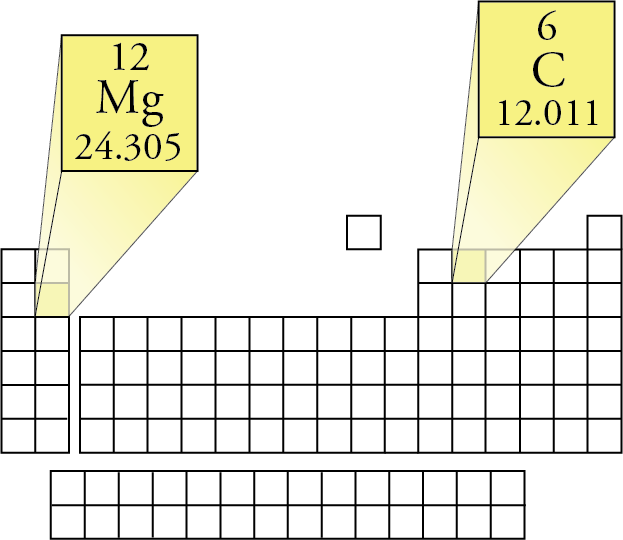 
Let me explain all this to you with the help of some examples: Multiply the number of atoms with the Ar value.Use the periodic table to find the A r value(s) of the element.To find the molar mass, we are going to do four simple steps: How to find the molar mass on the periodic table? With this, it is time to move on and talk about the calculation of molar mass. As you may know, these elements are arranged by (increasing) atomic number (number of protons in the nucleus of the atom). The periodic table of chemical elements is a table that displays the chemical elements. Similarly, the mass of 1 mole of H2O molecule is 18g.īut before telling you how to calculate the molar mass using the periodic table, let me tell you what a periodic table is. However, the numerical values of both of them are the same.įor example, the molecular mass of 1 H2O molecule is 18 a.m.u. Molecular mass (also known as molecular weight) is also the mass of one molecule of a substance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
